Sbírka Atom Meaning
Sbírka Atom Meaning. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.
Nejlepší 1
In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.
In electrically neutral atoms, the number of protons equals the number of electrons. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons.. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons... The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.
The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom... Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom... The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. In electrically neutral atoms, the number of protons equals the number of electrons. In electrically neutral atoms, the number of protons equals the number of electrons.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen... The smallest particle of an element that can exist either alone or in combination an atom of hydrogen... In electrically neutral atoms, the number of protons equals the number of electrons.

In electrically neutral atoms, the number of protons equals the number of electrons. In electrically neutral atoms, the number of protons equals the number of electrons.. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. In electrically neutral atoms, the number of protons equals the number of electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons... In electrically neutral atoms, the number of protons equals the number of electrons.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons... The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.
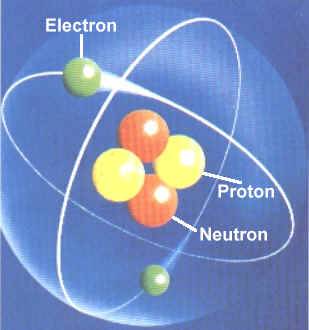
The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom... The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons.. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons... The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons.. In electrically neutral atoms, the number of protons equals the number of electrons.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom... The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. In electrically neutral atoms, the number of protons equals the number of electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom... Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.
/atomic-structure-artwork-549603139-57fe40e75f9b586c3537ebf4.jpg)
The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom... The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. In electrically neutral atoms, the number of protons equals the number of electrons.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom... Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.
/atom-57e1bb583df78c9cce33a106.jpg)
The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.. In electrically neutral atoms, the number of protons equals the number of electrons.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.. In electrically neutral atoms, the number of protons equals the number of electrons.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons.

In electrically neutral atoms, the number of protons equals the number of electrons. In electrically neutral atoms, the number of protons equals the number of electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.

In electrically neutral atoms, the number of protons equals the number of electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.

In electrically neutral atoms, the number of protons equals the number of electrons. In electrically neutral atoms, the number of protons equals the number of electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons.. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.
Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons... The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

In electrically neutral atoms, the number of protons equals the number of electrons.. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen... The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. In electrically neutral atoms, the number of protons equals the number of electrons. In electrically neutral atoms, the number of protons equals the number of electrons.

In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons... The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.

In electrically neutral atoms, the number of protons equals the number of electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. In electrically neutral atoms, the number of protons equals the number of electrons... The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.

The smallest particle of an element that can exist either alone or in combination an atom of hydrogen... Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.. In electrically neutral atoms, the number of protons equals the number of electrons.

The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom... The smallest particle of an element that can exist either alone or in combination an atom of hydrogen. In electrically neutral atoms, the number of protons equals the number of electrons. The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom. Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons... In electrically neutral atoms, the number of protons equals the number of electrons.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons. . The atom considered as a source of vast potential constructive or destructive energy … a largely forgotten legacy of this country's conquest of the atom.

Atoms remain intact in chemical reactions except for the removal, transfer, or exchange of certain electrons... . The smallest particle of an element that can exist either alone or in combination an atom of hydrogen.