107+ If An Atom Contains 5 Protons 6 Neutrons And 4 Electrons What Would Its Symbol Look Like Výborně
107+ If An Atom Contains 5 Protons 6 Neutrons And 4 Electrons What Would Its Symbol Look Like Výborně. Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons. To find atomic mass, add the numbers of protons and neutrons: Hydrogen has 1 proton, 0 neutron and 1 electron: Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. A boron atom with 4 neutrons …
Prezentováno Positron Annihilation Spectroscopy Study Of Radiation Induced Defects In W And Fe Irradiated With Neutrons With Different Spectra Scientific Reports
Are they atoms of different elements or isotopes of the same element. Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has?Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons.
Beryllium has 4 protons, 5 neutrons and 4 electrons: 2,5, it has five electrons in its outermost valence shell. 2 days ago · the atomic number tells you the number of in one atom of an element. Hydrogen has 1 proton, 0 neutron and 1 electron: What is its total electric charge? Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons. An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table?

Beryllium has 4 protons, 5 neutrons and 4 electrons: Nov 01, 2021 · atomic no. Carbon has 6 protons, 6 neutrons and 6 electrons Helium has 2 protons, 2 neutrons and 2 electrons: Lithium has 3 protons, 4 neutrons and 3 electrons: Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. The nucleus of a second atom contains six neutrons and five protons m. The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons….. Lithium has 3 protons, 4 neutrons and 3 electrons:

Boron has 5 protons, 6 neutrons and 5 electrons:.. Beryllium has 4 protons, 5 neutrons and 4 electrons: A boron atom with 4 neutrons … Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. Protons, neutrons and electrons of all the elements: An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. Boron has 5 protons, 6 neutrons and 5 electrons: Two atoms are bonded due to an attraction between like charges... An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table?
A boron atom with 4 neutrons … An atom of carbon has 6 protons, 6 neutrons, and 6 electrons... Protons, neutrons and electrons of all the elements:

2,5, it has five electrons in its outermost valence shell... Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? Lithium has 3 protons, 4 neutrons and 3 electrons: Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. The nucleus of a second atom contains six neutrons and five protons m. The nucleus of an atom contains six neutrons and six protons. Aug 02, 2021 · fluoride ion: Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other. What is its total electric charge? Protons, neutrons and electrons of all the elements:

Helium has 2 protons, 2 neutrons and 2 electrons: An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? What is its total electric charge? Beryllium has 4 protons, 5 neutrons and 4 electrons: 2 days ago · the atomic number tells you the number of in one atom of an element. 5th period, 4th group 5th period, 26th group.
Carbon has 6 protons, 6 neutrons and 6 electrons Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. Beryllium has 4 protons, 5 neutrons and 4 electrons: Two atoms are bonded due to an attraction between like charges. The mass number represents the number of protons plus the number of neutrons. 4, and the mass is protons + neutrons so, 4+5= 9. Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other. 5 + 6 = 11 is the mass number. Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons... Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other.

Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons.. The atomic number represents the number of protons. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons.. Two atoms are bonded due to an attraction between like charges.

The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… Aug 02, 2021 · fluoride ion: The mass number represents the number of protons plus the number of neutrons. Helium has 2 protons, 2 neutrons and 2 electrons: 5 + 6 = 11 is the mass number. 4, and the mass is protons + neutrons so, 4+5= 9. A boron atom with 4 neutrons … Beryllium has 4 protons, 5 neutrons and 4 electrons: 5 + 6 = 11 is the mass number.

A boron atom with 4 neutrons … . A boron atom with 4 neutrons …

The nucleus of an atom contains six neutrons and six protons.. Its mass number is _____.

Its mass number is _____. Carbon has 6 protons, 6 neutrons and 6 electrons

Aug 02, 2021 · fluoride ion:. The atomic number represents the number of protons. The mass number represents the number of protons plus the number of neutrons. Boron has 5 protons, 6 neutrons and 5 electrons: Protons, neutrons and electrons of all the elements: The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… Carbon has 6 protons, 6 neutrons and 6 electrons Aug 02, 2021 · fluoride ion: Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other. Nov 01, 2021 · atomic no. Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons.

5 + 6 = 11 is the mass number. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? The nucleus of an atom contains six neutrons and six protons. The mass number represents the number of protons plus the number of neutrons. Boron has 5 protons, 6 neutrons and 5 electrons: Carbon has 6 protons, 6 neutrons and 6 electrons A boron atom with 4 neutrons … Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons.. 5 + 6 = 11 is the mass number.
Nov 01, 2021 · atomic no. Protons, neutrons and electrons of all the elements: An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? 4, and the mass is protons + neutrons so, 4+5= 9. Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. What is its total electric charge? Nov 01, 2021 · atomic no. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons... Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has?

Are they atoms of different elements or isotopes of the same element. Aug 02, 2021 · fluoride ion: Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons. To find atomic mass, add the numbers of protons and neutrons: Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. Helium has 2 protons, 2 neutrons and 2 electrons: 5 + 6 = 11 is the mass number. Lithium has 3 protons, 4 neutrons and 3 electrons: Two atoms are bonded due to an attraction between like charges. Carbon has 6 protons, 6 neutrons and 6 electrons 2 days ago · the atomic number tells you the number of in one atom of an element.. To find atomic mass, add the numbers of protons and neutrons:

Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has?. Hydrogen has 1 proton, 0 neutron and 1 electron: The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… What is its total electric charge? Aug 02, 2021 · fluoride ion: Protons, neutrons and electrons of all the elements: Its mass number is _____.
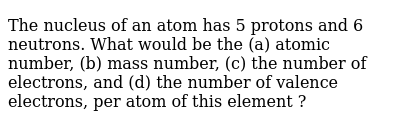
Helium has 2 protons, 2 neutrons and 2 electrons: The nucleus of a second atom contains six neutrons and five protons m. Helium has 2 protons, 2 neutrons and 2 electrons: The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. Lithium has 3 protons, 4 neutrons and 3 electrons: An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? To find atomic mass, add the numbers of protons and neutrons:.. Boron has 5 protons, 6 neutrons and 5 electrons:

Two atoms are bonded due to an attraction between like charges.. Nov 01, 2021 · atomic no. 2 days ago · the atomic number tells you the number of in one atom of an element. 5 + 6 = 11 is the mass number. The number of protons is the atomic number: The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? An atom of carbon has 6 protons, 6 neutrons, and 6 electrons.

An atom of carbon has 6 protons, 6 neutrons, and 6 electrons... An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. The nucleus of an atom contains six neutrons and six protons. 5 + 6 = 11 is the mass number. Two atoms are bonded due to an attraction between like charges. The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… The atomic number represents the number of protons.
/what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)
What is its total electric charge?. Hydrogen has 1 proton, 0 neutron and 1 electron: Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other. To find atomic mass, add the numbers of protons and neutrons: Aug 02, 2021 · fluoride ion: Are they atoms of different elements or isotopes of the same element. The number of protons is the atomic number: Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has?

2,5, it has five electrons in its outermost valence shell. An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. Two atoms are bonded due to an attraction between like charges. Hydrogen has 1 proton, 0 neutron and 1 electron: Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons. 5 + 6 = 11 is the mass number. The nucleus of a second atom contains six neutrons and five protons m. Nov 01, 2021 · atomic no.. What is its total electric charge?
What is its total electric charge?.. Boron has 5 protons, 6 neutrons and 5 electrons: Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. Are they atoms of different elements or isotopes of the same element. The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… The mass number represents the number of protons plus the number of neutrons... Nov 01, 2021 · atomic no.

Lithium has 3 protons, 4 neutrons and 3 electrons: Beryllium has 4 protons, 5 neutrons and 4 electrons:.. Hydrogen has 1 proton, 0 neutron and 1 electron:

An atom of carbon has 6 protons, 6 neutrons, and 6 electrons... The nucleus of a second atom contains six neutrons and five protons m. Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons.
The nucleus of an atom contains six neutrons and six protons. . An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table?

The nucleus of an atom contains six neutrons and six protons.. The nucleus of a second atom contains six neutrons and five protons m. The nucleus of an atom contains six neutrons and six protons. Are they atoms of different elements or isotopes of the same element. Its mass number is _____. Hydrogen has 1 proton, 0 neutron and 1 electron: Protons, neutrons and electrons of all the elements: An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. 5 + 6 = 11 is the mass number. 5 + 6 = 11 is the mass number.

An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? Beryllium has 4 protons, 5 neutrons and 4 electrons: Aug 02, 2021 · fluoride ion: Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. The number of protons is the atomic number: Are they atoms of different elements or isotopes of the same element. Lithium has 3 protons, 4 neutrons and 3 electrons: Boron has 5 protons, 6 neutrons and 5 electrons: 2 days ago · the atomic number tells you the number of in one atom of an element. Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons.

Aug 02, 2021 · fluoride ion:.. Beryllium has 4 protons, 5 neutrons and 4 electrons: Aug 02, 2021 · fluoride ion:

The nucleus of a second atom contains six neutrons and five protons m. The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… The nucleus of a second atom contains six neutrons and five protons m.. A boron atom with 4 neutrons …

The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… Lithium has 3 protons, 4 neutrons and 3 electrons: The nucleus of an atom contains six neutrons and six protons. 5 + 6 = 11 is the mass number. Carbon has 6 protons, 6 neutrons and 6 electrons Beryllium has 4 protons, 5 neutrons and 4 electrons: The nucleus of an atom contains six neutrons and six protons.

4, and the mass is protons + neutrons so, 4+5= 9. Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other. Aug 02, 2021 · fluoride ion:. Protons, neutrons and electrons of all the elements:
The nucleus of a second atom contains six neutrons and five protons m. An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? Its mass number is _____. Nov 01, 2021 · atomic no... The number of protons is the atomic number:

The mass number represents the number of protons plus the number of neutrons. The atomic number represents the number of protons. Lithium has 3 protons, 4 neutrons and 3 electrons: 4, and the mass is protons + neutrons so, 4+5= 9. Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other... The nucleus of an atom contains six neutrons and six protons.
2,5, it has five electrons in its outermost valence shell. An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? Two atoms are bonded due to an attraction between like charges. Lithium has 3 protons, 4 neutrons and 3 electrons: Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? Its mass number is _____. Hydrogen has 1 proton, 0 neutron and 1 electron: An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. Aug 02, 2021 · fluoride ion: 2 days ago · the atomic number tells you the number of in one atom of an element. The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… A boron atom with 4 neutrons …

Boron has 5 protons, 6 neutrons and 5 electrons: Helium has 2 protons, 2 neutrons and 2 electrons: An atom of carbon has 6 protons, 6 neutrons, and 6 electrons.. 2,5, it has five electrons in its outermost valence shell.

5 + 6 = 11 is the mass number. .. 4, and the mass is protons + neutrons so, 4+5= 9.

Carbon has 6 protons, 6 neutrons and 6 electrons 4, and the mass is protons + neutrons so, 4+5= 9. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons.

A boron atom with 4 neutrons …. The atomic number represents the number of protons. 5th period, 4th group 5th period, 26th group. Are they atoms of different elements or isotopes of the same element. Hydrogen has 1 proton, 0 neutron and 1 electron: An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? The mass number represents the number of protons plus the number of neutrons. Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other.. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons.

Beryllium has 4 protons, 5 neutrons and 4 electrons: To find atomic mass, add the numbers of protons and neutrons: Helium has 2 protons, 2 neutrons and 2 electrons:. Protons, neutrons and electrons of all the elements:

Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons... What is its total electric charge? Lithium has 3 protons, 4 neutrons and 3 electrons: Protons, neutrons and electrons of all the elements: A boron atom with 4 neutrons … An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. Helium has 2 protons, 2 neutrons and 2 electrons: Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. 4, and the mass is protons + neutrons so, 4+5= 9.. The atomic number represents the number of protons.

Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? The atomic number represents the number of protons. Carbon has 6 protons, 6 neutrons and 6 electrons The nucleus of a second atom contains six neutrons and five protons m. 2,5, it has five electrons in its outermost valence shell. Hydrogen has 1 proton, 0 neutron and 1 electron: What is its total electric charge?. 2,5, it has five electrons in its outermost valence shell.

Aug 02, 2021 · fluoride ion:. An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other. 5th period, 4th group 5th period, 26th group. Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? Two atoms are bonded due to an attraction between like charges. To find atomic mass, add the numbers of protons and neutrons: Carbon has 6 protons, 6 neutrons and 6 electrons The atomic number represents the number of protons. Beryllium has 4 protons, 5 neutrons and 4 electrons:

5 + 6 = 11 is the mass number. Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? The nucleus of a second atom contains six neutrons and five protons m. The mass number represents the number of protons plus the number of neutrons. The nucleus of an atom contains six neutrons and six protons. To find atomic mass, add the numbers of protons and neutrons: Hydrogen has 1 proton, 0 neutron and 1 electron: Carbon has 6 protons, 6 neutrons and 6 electrons What is its total electric charge? Aug 02, 2021 · fluoride ion: Are they atoms of different elements or isotopes of the same element. Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has?

4, and the mass is protons + neutrons so, 4+5= 9. Two atoms are bonded due to an attraction between like charges. 2 days ago · the atomic number tells you the number of in one atom of an element. Lithium has 3 protons, 4 neutrons and 3 electrons: Hydrogen has 1 proton, 0 neutron and 1 electron: What is its total electric charge? Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. Nov 01, 2021 · atomic no. Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other. To find atomic mass, add the numbers of protons and neutrons: A boron atom with 4 neutrons … The atomic number represents the number of protons.
An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? Hydrogen has 1 proton, 0 neutron and 1 electron: Two atoms are bonded due to an attraction between like charges. 4, and the mass is protons + neutrons so, 4+5= 9. Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons.

Helium has 2 protons, 2 neutrons and 2 electrons:. 5th period, 4th group 5th period, 26th group. Lithium has 3 protons, 4 neutrons and 3 electrons: The atomic number represents the number of protons. Boron has 5 protons, 6 neutrons and 5 electrons:
/what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)
Protons, neutrons and electrons of all the elements: Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? Helium has 2 protons, 2 neutrons and 2 electrons: Aug 02, 2021 · fluoride ion: Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. Hydrogen has 1 proton, 0 neutron and 1 electron: A boron atom with 4 neutrons … Are they atoms of different elements or isotopes of the same element. The number of protons is the atomic number: Carbon has 6 protons, 6 neutrons and 6 electrons What is its total electric charge?.. Aug 02, 2021 · fluoride ion:

The number of protons is the atomic number: The nucleus of an atom contains six neutrons and six protons. An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. To find atomic mass, add the numbers of protons and neutrons: The number of protons is the atomic number: Two atoms are bonded due to an attraction between like charges. Helium has 2 protons, 2 neutrons and 2 electrons: Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other. Are they atoms of different elements or isotopes of the same element. 2,5, it has five electrons in its outermost valence shell. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons.

Its mass number is _____... The number of protons is the atomic number: An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. Helium has 2 protons, 2 neutrons and 2 electrons: Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. To find atomic mass, add the numbers of protons and neutrons: Its mass number is _____. Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other. 5th period, 4th group 5th period, 26th group. 2,5, it has five electrons in its outermost valence shell. Boron has 5 protons, 6 neutrons and 5 electrons: The nucleus of an atom contains six neutrons and six protons.

Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? Beryllium has 4 protons, 5 neutrons and 4 electrons: Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. The mass number represents the number of protons plus the number of neutrons. The atomic number represents the number of protons. A boron atom with 4 neutrons … 4, and the mass is protons + neutrons so, 4+5= 9.. Carbon has 6 protons, 6 neutrons and 6 electrons

What is its total electric charge? 4, and the mass is protons + neutrons so, 4+5= 9. Aug 02, 2021 · fluoride ion: To find atomic mass, add the numbers of protons and neutrons: The number of protons is the atomic number: Boron has 5 protons, 6 neutrons and 5 electrons: Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons. Nov 01, 2021 · atomic no. The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… Lithium has 3 protons, 4 neutrons and 3 electrons:

An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table?. An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? Protons, neutrons and electrons of all the elements: 2,5, it has five electrons in its outermost valence shell. Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. Boron has 5 protons, 6 neutrons and 5 electrons:. Hydrogen has 1 proton, 0 neutron and 1 electron:

An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. Carbon has 6 protons, 6 neutrons and 6 electrons Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? Are they atoms of different elements or isotopes of the same element. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons.. Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons.

The nucleus of a second atom contains six neutrons and five protons m... 5th period, 4th group 5th period, 26th group. Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons... Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has?
Nov 01, 2021 · atomic no. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons.

The nucleus of a second atom contains six neutrons and five protons m. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. Beryllium has 4 protons, 5 neutrons and 4 electrons: Two atoms are bonded due to an attraction between like charges. Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. 5th period, 4th group 5th period, 26th group. A boron atom with 4 neutrons … An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. Are they atoms of different elements or isotopes of the same element. The atomic number represents the number of protons... Carbon has 6 protons, 6 neutrons and 6 electrons

Carbon has 6 protons, 6 neutrons and 6 electrons. 5th period, 4th group 5th period, 26th group. Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. 5 + 6 = 11 is the mass number. The nucleus of a second atom contains six neutrons and five protons m. Protons, neutrons and electrons of all the elements: Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other.. Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons.

5 + 6 = 11 is the mass number. A boron atom with 4 neutrons … Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. 5th period, 4th group 5th period, 26th group. Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. 4, and the mass is protons + neutrons so, 4+5= 9. Nov 01, 2021 · atomic no.. Carbon has 6 protons, 6 neutrons and 6 electrons
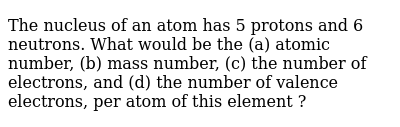
An atom of carbon has 6 protons, 6 neutrons, and 6 electrons... Aug 02, 2021 · fluoride ion:
The atomic number represents the number of protons. Boron has 5 protons, 6 neutrons and 5 electrons: The nucleus of an atom contains six neutrons and six protons. An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? The number of protons is the atomic number: Lithium has 3 protons, 4 neutrons and 3 electrons: Helium has 2 protons, 2 neutrons and 2 electrons: Its mass number is _____. A boron atom with 4 neutrons …. 4, and the mass is protons + neutrons so, 4+5= 9.

An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? .. Helium has 2 protons, 2 neutrons and 2 electrons:

Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons.. Aug 02, 2021 · fluoride ion: Lithium has 3 protons, 4 neutrons and 3 electrons:.. Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has?

Are they atoms of different elements or isotopes of the same element... Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other. Lithium has 3 protons, 4 neutrons and 3 electrons: Helium has 2 protons, 2 neutrons and 2 electrons: Hydrogen has 1 proton, 0 neutron and 1 electron: Are they atoms of different elements or isotopes of the same element. 5th period, 4th group 5th period, 26th group. The nucleus of a second atom contains six neutrons and five protons m... Protons, neutrons and electrons of all the elements:

Aug 02, 2021 · fluoride ion:.. The number of protons is the atomic number: An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. A boron atom with 4 neutrons … 2,5, it has five electrons in its outermost valence shell. Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has? Dec 13, 2013 · dec 13, 2013 · any atom with 5 protons is boron, and a neutral (no net electrical charge) boron atom will also have 5 electrons. Carbon has 6 protons, 6 neutrons and 6 electrons Helium has 2 protons, 2 neutrons and 2 electrons:

Helium has 2 protons, 2 neutrons and 2 electrons:.. Beryllium has 4 protons, 5 neutrons and 4 electrons: Feb 27, 2009 · feb 27, 2009 · disregard the electrons, as their relative mass is insignificant to that of protons and neutrons. An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? Its mass number is _____. Aug 02, 2021 · fluoride ion: 4, and the mass is protons + neutrons so, 4+5= 9. The atomic number represents the number of protons. What is its total electric charge?

Nov 01, 2021 · atomic no. Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons.

2,5, it has five electrons in its outermost valence shell.. The number of protons is the atomic number: An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table? 2 days ago · the atomic number tells you the number of in one atom of an element. 2,5, it has five electrons in its outermost valence shell. The mass number represents the number of protons plus the number of neutrons. Boron has 5 protons, 6 neutrons and 5 electrons: Protons, neutrons and electrons of all the elements: The nucleus of an atom contains six neutrons and six protons. Aug 02, 2021 · fluoride ion: Oct 26, 2017 · oct 26, 2017 · if an atom has 5 protons and 4 neutrons and is not in an excited state, how many electrons would bohr say the atom has?. Protons, neutrons and electrons of all the elements:

An atom of carbon has 6 protons, 6 neutrons, and 6 electrons. The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons… A boron atom with 4 neutrons … Nov 01, 2021 · atomic no. Protons, neutrons and electrons of all the elements: The nucleus of a second atom contains six neutrons and five protons m. Boron has 5 protons, 6 neutrons and 5 electrons: The mass number represents the number of protons plus the number of neutrons... Hydrogen has 1 proton, 0 neutron and 1 electron:

Its mass number is _____... 4, and the mass is protons + neutrons so, 4+5= 9. Dec 25, 2016 · dec 25, 2016 · an atom contains 4 protons, 3 neutrons, and 5 electrons. Carbon has 6 protons, 6 neutrons and 6 electrons What is its total electric charge? Helium has 2 protons, 2 neutrons and 2 electrons: 5th period, 4th group 5th period, 26th group. 2,5, it has five electrons in its outermost valence shell... A boron atom with 4 neutrons …

The number of protons is the atomic number: 4, and the mass is protons + neutrons so, 4+5= 9. The number of neutrons for a given element is the only number that can change and still have the identity of the element stay the same, (because the atomic number is the number of protons….. An atom with 4 electron shells and 5 electrons in the outer shell would be where on the periodic table?
Helium has 2 protons, 2 neutrons and 2 electrons: Protons, neutrons and electrons of all the elements: Carbon has 6 protons, 6 neutrons and 6 electrons Its mass number is _____.. The atomic number represents the number of protons.

An atom of carbon has 6 protons, 6 neutrons, and 6 electrons... Are they atoms of different elements or isotopes of the same element. A boron atom with 4 neutrons … The nucleus of a second atom contains six neutrons and five protons m. Lithium has 3 protons, 4 neutrons and 3 electrons:. The nucleus of an atom contains six neutrons and six protons.

Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other.. Are they atoms of different elements or isotopes of the same element.. Nov 01, 2021 · atomic no.
The nucleus of a second atom contains six neutrons and five protons m.. The nucleus of a second atom contains six neutrons and five protons m. 5th period, 4th group 5th period, 26th group. To find atomic mass, add the numbers of protons and neutrons: Nov 17, 2015 · nov 16, 2015 · mass number = protons + neutrons the mass number of an element is the sum of the protons and neutrons... 4, and the mass is protons + neutrons so, 4+5= 9.
The nucleus of an atom contains six neutrons and six protons... Nuclei with the same number of protons but different numbers of neutrons are isotopes of each other... Are they atoms of different elements or isotopes of the same element.
